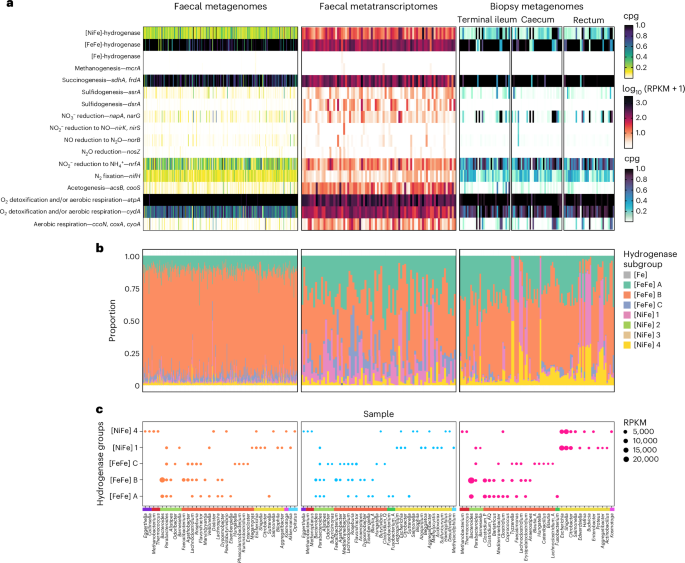
A widespread hydrogenase supports fermentative growth of gut bacteria in healthy people
Cummings, J. & Macfarlane, G. The control and consequences of bacterial fermentation in the human colon. J. Appl. Bacteriol. 70, 443–459 (1991).
Google Scholar
Hylemon, P. B., Harris, S. C. & Ridlon, J. M. Metabolism of hydrogen gases and bile acids in the gut microbiome. FEBS Lett. 592, 2070–2082 (2018).
Google Scholar
Nakamura, N., Lin, H. C., McSweeney, C. S., Mackie, R. I. & Gaskins, H. R. Mechanisms of microbial hydrogen disposal in the human colon and implications for health and disease. Annu. Rev. Food Sci. Technol. 1, 363–395 (2010).
Google Scholar
Kalantar-Zadeh, K., Berean, K. J., Burgell, R. E., Muir, J. G. & Gibson, P. R. Intestinal gases: influence on gut disorders and the role of dietary manipulations. Nat. Rev. Gastroenterol. Hepatol. 16, 733–747 (2019).
Google Scholar
Oliphant, K. & Allen-Vercoe, E. Macronutrient metabolism by the human gut microbiome: major fermentation by-products and their impact on host health. Microbiome 7, 91 (2019).
Google Scholar
Carbonero, F., Benefiel, A. C. & Gaskins, H. R. Contributions of the microbial hydrogen economy to colonic homeostasis. Nat. Rev. Gastroenterol. Hepatol. 9, 504–518 (2012).
Google Scholar
Pimentel, M., Mathur, R. & Chang, C. Gas and the microbiome. Curr. Gastroenterol. Rep. 15, 356 (2013).
Google Scholar
Christl, S. U., Murgatroyd, P. R., Gibson, G. R. & Cummings, J. H. Production, metabolism, and excretion of hydrogen in the large intestine. Gastroenterology 102, 1269–1277 (1992).
Google Scholar
Levitt, M. D. Production and excretion of hydrogen gas in man. N. Engl. J. Med. 281, 122–127 (1969).
Google Scholar
Suarez, F., Furne, J., Springfield, J. & Levitt, M. Insights into human colonic physiology obtained from the study of flatus composition. Am. J. Physiol. 272, G1028–G1033 (1997).
Google Scholar
Christl, S., Gibson, G. & Cummings, J. Role of dietary sulphate in the regulation of methanogenesis in the human large intestine. Gut 33, 1234–1238 (1992).
Google Scholar
Gibson, G. et al. Alternative pathways for hydrogen disposal during fermentation in the human colon. Gut 31, 679–683 (1990).
Google Scholar
Gibson, G., Macfarlane, G. & Cummings, J. Sulphate reducing bacteria and hydrogen metabolism in the human large intestine. Gut 34, 437–439 (1993).
Google Scholar
Smith, N. W., Shorten, P. R., Altermann, E. H., Roy, N. C. & McNabb, W. C. Hydrogen cross-feeders of the human gastrointestinal tract. Gut Microbes 10, 270–288 (2019).
Google Scholar
Bryant, M., Wolin, E., Wolin, M. & Wolfe, R. Methanobacillus omelianskii, a symbiotic association of two species of bacteria. Archiv. Mikrobiol 59, 20–31 (1967).
Google Scholar
Dolfing, J. Syntrophy in microbial fuel cells. ISME J. 8, 4–5 (2014).
Google Scholar
Greening, C. et al. Diverse hydrogen production and consumption pathways influence methane production in ruminants. ISME J. 13, 2617–2632 (2019).
Google Scholar
Strocchi, A. & Levitt, M. D. Factors affecting hydrogen production and consumption by human fecal flora. The critical roles of hydrogen tension and methanogenesis. J. Clin. Invest. 89, 1304–1311 (1992).
Google Scholar
Wolf, P. G., Biswas, A., Morales, S. E., Greening, C. & Gaskins, H. R. H2 metabolism is widespread and diverse among human colonic microbes. Gut Microbes 7, 235–245 (2016).
Google Scholar
Ruaud, A. et al. Syntrophy via interspecies H2 transfer between Christensenella and Methanobrevibacter underlies their global cooccurrence in the human gut. mBio 11, e03235-19 (2020).
Campbell, A., Gdanetz, K., Schmidt, A. W. & Schmidt, T. M. H2 generated by fermentation in the human gut microbiome influences metabolism and competitive fitness of gut butyrate producers. Microbiome 11, 133 (2023).
Google Scholar
Rey, F. E. et al. Metabolic niche of a prominent sulfate-reducing human gut bacterium. Proc. Natl Acad. Sci. USA 110, 13582–13587 (2013).
Google Scholar
McCurry, M. D. et al. Gut bacteria convert glucocorticoids into progestins in the presence of hydrogen gas. Cell 187, 2952–2968.e13 (2024).
Ge, L., Yang, M., Yang, N.-N., Yin, X.-X. & Song, W.-G. Molecular hydrogen: a preventive and therapeutic medical gas for various diseases. Oncotarget 8, 102653–102673 (2017).
Google Scholar
Ohta, S. Recent progress toward hydrogen medicine: potential of molecular hydrogen for preventive and therapeutic applications. Curr. Pharm. Des. 17, 2241–2252 (2011).
Google Scholar
King, T., Elia, M. & Hunter, J. Abnormal colonic fermentation in irritable bowel syndrome. Lancet 352, 1187–1189 (1998).
Google Scholar
Metz, G., Peters, T., Jenkins, D. A., Newman, A. & Blendis, L. Breath hydrogen as a diagnostic method for hypolactasia. Lancet 305, 1155–1157 (1975).
Google Scholar
Rhodes, J., Middleton, P. & Jewell, D. The lactulose hydrogen breath test as a diagnostic test for small-bowel bacterial overgrowth. Scand. J. Gastroenterol. 14, 333–336 (1979).
Google Scholar
Simrén, M. & Stotzer, P. Use and abuse of hydrogen breath tests. Gut 55, 297–303 (2006).
Google Scholar
Benoit, S. L., Maier, R. J., Sawers, R. G. & Greening, C. Molecular hydrogen metabolism: a widespread trait of pathogenic bacteria and protists. Microbiol. Mol. Biol. Rev. 84, e00092-19 (2020).
Hughes, E. R. et al. Reshaping of bacterial molecular hydrogen metabolism contributes to the outgrowth of commensal E. coli during gut inflammation. eLife 10, e58609 (2021).
Google Scholar
Maier, L. et al. Microbiota-derived hydrogen fuels Salmonella typhimurium invasion of the gut ecosystem. Cell Host Microbe 14, 641–651 (2013).
Google Scholar
Olson, J. W. & Maier, R. J. Molecular hydrogen as an energy source for Helicobacter pylori. Science 298, 1788–1790 (2002).
Google Scholar
Weerakoon, D. R., Borden, N. J., Goodson, C. M., Grimes, J. & Olson, J. W. The role of respiratory donor enzymes in Campylobacter jejuni host colonization and physiology. Microb. Pathog. 47, 8–15 (2009).
Google Scholar
Kaji, M. et al. The hydA gene encoding the H2-evolving hydrogenase of Clostridium perfringens: molecular characterization and expression of the gene. FEMS Microbiol. Lett. 181, 329–336 (1999).
Google Scholar
Lloyd, D., Ralphs, J. R. & Harris, J. C. Giardia intestinalis, a eukaryote without hydrogenosomes, produces hydrogen. Microbiology 148, 727–733 (2002).
Google Scholar
Greening, C. et al. Genomic and metagenomic surveys of hydrogenase distribution indicate H2 is a widely utilised energy source for microbial growth and survival. ISME J. 10, 761–777 (2016).
Google Scholar
Vignais, P. M. in Bioenergetics: Energy Conservation and Conversion (eds Schäfer, G. & Penefsky, H. S.) 223–252 (Springer, 2007).
Lubitz, W., Ogata, H., Rudiger, O. & Reijerse, E. Hydrogenases. Chem. Rev. 114, 4081–4148 (2014).
Google Scholar
Peters, J. W., Lanzilotta, W. N., Lemon, B. J. & Seefeldt, L. C. X-ray crystal structure of the Fe-only hydrogenase (CpI) from Clostridium pasteurianum to 1.8 angstrom resolution. Science 282, 1853–1858 (1998).
Google Scholar
McDowall, J. S. et al. Bacterial formate hydrogenlyase complex. Proc. Natl Acad. Sci. USA 111, E3948–E3956 (2014).
Google Scholar
Steinhilper, R., Höff, G., Heider, J. & Murphy, B. J. Structure of the membrane-bound formate hydrogenlyase complex from Escherichia coli. Nat. Commun. 13, 5395 (2022).
Google Scholar
Calusinska, M., Happe, T., Joris, B. & Wilmotte, A. The surprising diversity of clostridial hydrogenases: a comparative genomic perspective. Microbiology 156, 1575–1588 (2010).
Google Scholar
Fasano, A., Jacq-Bailly, A., Wozniak, J., Fourmond, V. & Léger, C. Catalytic bias and redox-driven inactivation of the group B FeFe hydrogenase CpIII. ACS Catal. 14, 7001–7010 (2024).
Google Scholar
Greening, C. et al. Minimal and hybrid hydrogenases are active from archaea. Cell 187, 3357–3372.e19 (2024).
Furlan, C. et al. Structural insight on the mechanism of an electron-bifurcating [FeFe] hydrogenase. eLife 11, e79361 (2022).
Google Scholar
Schuchmann, K. & Müller, V. A bacterial electron-bifurcating hydrogenase. J. Biol. Chem. 287, 31165–31171 (2012).
Google Scholar
Wang, S., Huang, H., Kahnt, J. & Thauer, R. K. A reversible electron-bifurcating ferredoxin-and NAD-dependent [FeFe]-hydrogenase (HydABC) in Moorella thermoacetica. J. Bacteriol. 195, 1267–1275 (2013).
Google Scholar
Zheng, Y., Kahnt, J., Kwon, I. H., Mackie, R. I. & Thauer, R. K. Hydrogen formation and its regulation in Ruminococcus albus: involvement of an electron-bifurcating [FeFe]-hydrogenase, of a non-electron-bifurcating [FeFe]-hydrogenase, and of a putative hydrogen-sensing [FeFe]-hydrogenase. J. Bacteriol. 196, 3840–3852 (2014).
Google Scholar
Li, Q. S. et al. Dietary selection of metabolically distinct microorganisms drives hydrogen metabolism in ruminants. ISME J. 16, 2535–2546 (2022).
Google Scholar
Xie, F. et al. An integrated gene catalog and over 10,000 metagenome-assembled genomes from the gastrointestinal microbiome of ruminants. Microbiome 9, 137 (2021).
Google Scholar
McKay, L. F., Eastwood, M. & Brydon, W. Methane excretion in man—a study of breath, flatus, and faeces. Gut 26, 69–74 (1985).
Google Scholar
Miller, T. L. & Wolin, M. Methanogens in human and animal intestinal tracts. Syst. Appl. Microbiol. 7, 223–229 (1986).
Google Scholar
Braccia, D. J., Jiang, X., Pop, M. & Hall, A. B. The capacity to produce hydrogen sulfide (H2S) via cysteine degradation is ubiquitous in the human gut microbiome. Front. Microbiol. 12, 705583 (2021).
Google Scholar
Marcelino, V. R. et al. Disease-specific loss of microbial cross-feeding interactions in the human gut. Nat. Commun. 14, 6546 (2023).
Google Scholar
Wolf, P. G. et al. Diversity and distribution of sulfur metabolic genes in the human gut microbiome and their association with colorectal cancer. Microbiome 10, 64 (2022).
Google Scholar
Schirmer, M. et al. Dynamics of metatranscription in the inflammatory bowel disease gut microbiome. Nat. Microbiol. 3, 337–346 (2018).
Google Scholar
Cabrol, L. et al. Microbial ecology of fermentative hydrogen producing bioprocesses: useful insights for driving the ecosystem function. FEMS Microbiol. Rev. 41, 158–181 (2017).
Google Scholar
Kelly, W. J. et al. Hydrogen and formate production and utilisation in the rumen and the human colon. Anim. Microbiome 4, 22 (2022).
Google Scholar
Bulen, W. & LeComte, J. The nitrogenase system from Azotobacter: two-enzyme requirement for N2 reduction, ATP-dependent H2 evolution, and ATP hydrolysis. Proc. Natl Acad. Sci. USA 56, 979–986 (1966).
Google Scholar
Søndergaard, D., Pedersen, C. N. & Greening, C. HydDB: a web tool for hydrogenase classification and analysis. Sci. Rep. 6, 34212 (2016).
Google Scholar
Ballantine, S. P. & Boxer, D. H. Nickel-containing hydrogenase isoenzymes from anaerobically grown Escherichia coli K-12. J. Bacteriol. 163, 454–459 (1985).
Google Scholar
Lamichhane-Khadka, R., Kwiatkowski, A. & Maier, R. J. The Hyb hydrogenase permits hydrogen-dependent respiratory growth of Salmonella enterica serovar Typhimurium. mBio 1, e00284-10 (2010).
Katsyv, A. et al. Molecular basis of the electron bifurcation mechanism in the [FeFe]-hydrogenase complex HydABC. J. Am. Chem. Soc. 145, 5696–5709 (2023).
Google Scholar
Jones, R. B. et al. Inter-niche and inter-individual variation in gut microbial community assessment using stool, rectal swab, and mucosal samples. Sci. Rep. 8, 4139 (2018).
Google Scholar
Kim, D. et al. Comparison of sampling methods in assessing the microbiome from patients with ulcerative colitis. BMC Gastroenterol. 21, 396 (2021).
Google Scholar
Vaga, S. et al. Compositional and functional differences of the mucosal microbiota along the intestine of healthy individuals. Sci. Rep. 10, 14977 (2020).
Google Scholar
D’Adamo, G. L. et al. Bacterial clade-specific analysis identifies distinct epithelial responses in inflammatory bowel disease. Cell Rep. Med. 4, 101124 (2023).
Rossmann, R., Sawers, G. & Böck, A. Mechanism of regulation of the formate-hydrogenlyase pathway by oxygen, nitrate, and pH: definition of the formate regulon. Mol. Microbiol. 5, 2807–2814 (1991).
Google Scholar
Chassard, C., Delmas, E., Lawson, P. A. & Bernalier-Donadille, A. Bacteroides xylanisolvens sp. nov., a xylan-degrading bacterium isolated from human faeces. Int. J. Syst. Evol. Microbiol. 58, 1008–1013 (2008).
Harding, G., Sutter, V., Finegold, S. & Bricknell, K. Characterization of Bacteroides melaninogenicus. J. Clin. Microbiol. 4, 354–359 (1976).
Google Scholar
Kazimierowicz, J., Dębowski, M. & Zieliński, M. Effectiveness of hydrogen production by Bacteroides vulgatus in psychrophilic fermentation of cattle slurry. Clean Technol. 4, 806–814 (2022).
Google Scholar
Suzuki, A. et al. Quantification of hydrogen production by intestinal bacteria that are specifically dysregulated in Parkinson’s disease. PLoS ONE 13, e0208313 (2018).
Google Scholar
Adamska-Venkatesh, A. et al. Artificially maturated [FeFe] hydrogenase from Chlamydomonas reinhardtii: a HYSCORE and ENDOR study of a non-natural H-cluster. Phys. Chem. Chem. Phys. 17, 5421–5430 (2015).
Berggren, G. et al. Biomimetic assembly and activation of [FeFe]-hydrogenases. Nature 499, 66–69 (2013).
Google Scholar
Fischbach, M. A. & Sonnenburg, J. L. Eating for two: how metabolism establishes interspecies interactions in the gut. Cell Host Microbe 10, 336–347 (2011).
Google Scholar
Macy, J. M., Ljungdahl, L. G. & Gottschalk, G. Pathway of succinate and propionate formation in Bacteroides fragilis. J. Bacteriol. 134, 84–91 (1978).
Google Scholar
Miller, T. L. The pathway of formation of acetate and succinate from pyruvate by Bacteroides succinogenes. Arch. Microbiol. 117, 145–152 (1978).
Google Scholar
Pan, N. & Imlay, J. A. How does oxygen inhibit central metabolism in the obligate anaerobe Bacteroides thetaiotaomicron. Mol. Microbiol. 39, 1562–1571 (2001).
Google Scholar
Qu, Z. et al. Selective utilization of medicinal polysaccharides by human gut Bacteroides and Parabacteroides species. Nat. Commun. 16, 638 (2025).
Google Scholar
Baughn, A. D. & Malamy, M. H. The essential role of fumarate reductase in haem-dependent growth stimulation of Bacteroides fragilis. Microbiology 149, 1551–1558 (2003).
Google Scholar
Macy, J., Probst, I. & Gottschalk, G. Evidence for cytochrome involvement in fumarate reduction and adenosine 5′-triphosphate synthesis by Bacteroides fragilis grown in the presence of hemin. J. Bacteriol. 123, 436–442 (1975).
Google Scholar
Sperry, J., Appleman, M. & Wilkins, T. D. Requirement of heme for growth of Bacteroides fragilis. Appl. Environ. Microbiol. 34, 386–390 (1977).
Google Scholar
Hess, V. et al. Occurrence of ferredoxin: NAD+ oxidoreductase activity and its ion specificity in several Gram-positive and Gram-negative bacteria. PeerJ 4, e1515 (2016).
Google Scholar
Zhang, B., Lingga, C., De Groot, H. & Hackmann, T. J. The oxidoreductase activity of Rnf balances redox cofactors during fermentation of glucose to propionate in Prevotella. Sci. Rep. 13, 16429 (2023).
Google Scholar
Liu, H. et al. Functional genetics of human gut commensal Bacteroides thetaiotaomicron reveals metabolic requirements for growth across environments. Cell Rep. 34, 108789 (2021).
He, Q. et al. Two distinct metacommunities characterize the gut microbiota in Crohn’s disease patients. GigaScience (2017).
Little, A. S. et al. Dietary-and host-derived metabolites are used by diverse gut bacteria for anaerobic respiration. Nat. Microbiol. 9, 55–69 (2024).
Google Scholar
Andrews, S. FastQC v.0.11.7 (Babraham Bioinformatics, Babraham Institute, 2010).
Ewels, P., Magnusson, M., Lundin, S. & Käller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048 (2016).
Google Scholar
Bushnell, B. BBMap: a fast, accurate, splice-aware aligner (Lawrence Berkeley National Lab, 2014).
Kopylova, E., Noé, L. & Touzet, H. SortMeRNA: fast and accurate filtering of ribosomal RNAs in metatranscriptomic data. Bioinformatics 28, 3211–3217 (2012).
Google Scholar
Buchfink, B., Xie, C. & Huson, D. H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 12, 59–60 (2015).
Google Scholar
Leung, P. M. & Greening, C. Greening lab metabolic marker gene databases. Monash Univ. (2020).
Blanco-Míguez, A. et al. Extending and improving metagenomic taxonomic profiling with uncharacterized species using MetaPhlAn 4. Nat. Biotechnol. 41, 1633–1644 (2023).
Google Scholar
Forster, S. C. et al. A human gut bacterial genome and culture collection for improved metagenomic analyses. Nat. Biotechnol. 37, 186–192 (2019).
Google Scholar
Parks, D. H., Imelfort, M., Skennerton, C. T., Hugenholtz, P. & Tyson, G. W. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 25, 1043–1055 (2015).
Google Scholar
Chaumeil, P.-A., Mussig, A. J., Hugenholtz, P. & Parks, D. H. GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics 36, 1925–1927 (2019).
Letunic, I. & Bork, P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 49, W293–W296 (2021).
Google Scholar
Morra, S., Mongili, B., Maurelli, S., Gilardi, G. & Valetti, F. Isolation and characterization of a new [FeFe]-hydrogenase from Clostridium perfringens. Biotechnol. Appl. Biochem. 63, 305–311 (2016).
Google Scholar
Duncan, S. H., Hold, G. L., Harmsen, H. J., Stewart, C. S. & Flint, H. J. Growth requirements and fermentation products of Fusobacterium prausnitzii, and a proposal to reclassify it as Faecalibacterium prausnitzii gen. nov., comb. nov. Int. J. Syst. Evol. Microbiol. 52, 2141–2146 (2002).
Islam, Z. F. et al. Two Chloroflexi classes independently evolved the ability to persist on atmospheric hydrogen and carbon monoxide. ISME J. 13, 1801–1813 (2019).
Google Scholar
Albert, D. B. & Martens, C. S. Determination of low-molecular-weight organic acid concentrations in seawater and pore-water samples via HPLC. Mar. Chem. 56, 27–37 (1997).
Google Scholar
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Google Scholar
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Google Scholar
Shaffer, M. et al. DRAM for distilling microbial metabolism to automate the curation of microbiome function. Nucleic Acids Res. 48, 8883–8900 (2020).
Google Scholar
Kanehisa, M. & Goto, S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Google Scholar
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Google Scholar
Evans, R. et al. Protein complex prediction with AlphaFold-Multimer. Preprint at bioRxiv (2022).
Mirdita, M. et al. ColabFold: making protein folding accessible to all. Nat. Methods 19, 679–682 (2022).
Google Scholar
van Kempen, M. et al. Fast and accurate protein structure search with Foldseek. Nat. Biotechnol. 42, 243–246 (2023).
Duan, J. et al. Cyanide binding to [FeFe]-hydrogenase stabilizes the alternative configuration of the proton transfer pathway. Angew. Chem. Int. Ed. 62, e202216903 (2023).
Google Scholar
TranQui, D. & Jesior, J. Structure of the ferredoxin from Clostridium acidurici: model at 1.8 Å resolution. Acta Crystallogr. D 51, 155–159 (1995).
Google Scholar
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
Google Scholar
Land, H. et al. Discovery of novel [FeFe]-hydrogenases for biocatalytic H2-production. Chem. Sci. 10, 9941–9948 (2019).
Google Scholar
Li, H. & Rauchfuss, T. B. Iron carbonyl sulfides, formaldehyde, and amines condense to give the proposed azadithiolate cofactor of the Fe-only hydrogenases. J. Am. Chem. Soc. 124, 726–727 (2002).
Google Scholar
Zaffaroni, R., Rauchfuss, T. B., Gray, D. L., De Gioia, L. & Zampella, G. Terminal vs bridging hydrides of diiron dithiolates: protonation of Fe2(dithiolate)(CO)2(PMe3)4. J. Am. Chem. Soc. 134, 19260–19269 (2012).
Google Scholar
Khanna, N., Esmieu, C., Mészáros, L. S., Lindblad, P. & Berggren, G. In vivo activation of an [FeFe] hydrogenase using synthetic cofactors. Energy Environ. Sci. 10, 1563–1567 (2017).
Google Scholar
Le Cloirec, A. et al. A di-iron dithiolate possessing structural elements of the carbonyl/cyanide sub-site of the H-centre of Fe-only hydrogenase. Chem. Commun. 1999, 2285–2286 (1999).
Google Scholar
Lyon, E. J., Georgakaki, I. P., Reibenspies, J. H. & Darensbourg, M. Y. Carbon monoxide and cyanide ligands in a classical organometallic complex model for Fe-only hydrogenase. Angew. Chem. Int. Ed. 38, 3178–3180 (1999).
Google Scholar
Schmidt, M., Contakes, S. M. & Rauchfuss, T. B. First generation analogues of the binuclear site in the Fe-only hydrogenases: Fe2 (µ-SR)2(CO)4(CN)22−. J. Am. Chem. Soc. 121, 9736–9737 (1999).
Google Scholar
Krueger, F. Trim Galore!: a wrapper around Cutadapt and FastQC to consistently apply adapter and quality trimming to FastQ files, with extra functionality for RRBS data (Babraham Institute, 2015).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Google Scholar
Shen, W., Le, S., Li, Y. & Hu, F. SeqKit: a cross-platform and ultrafast toolkit for FASTA/Q file manipulation. PLoS ONE 11, e0163962 (2016).
Google Scholar
link







